This blog is intended for students in the health sciences and other students with an interest in cardiovascular, pulmonary and renal physiology and pathophysiology. It is a compilation of original contributions as well as notes I have taken during lectures on these topics and clinical lectures. At the bottom of each post is a box for comments that you are invited to use. Steve Wood, PhD, swood60@gmail.com teaching website: http://www.cvpulmrenal.com
Friday, May 3, 2013
Wednesday, May 1, 2013
World Record Metabolic Acidosis
A 44-year-old woman drank approximately 720 ml of ethylene glycol in the form of antifreeze. She had previously attempted suicide by injecting the same substance into her buttocks. When admitted to the hospital, the patient was unresponsive and incontinent and was receiving ventilation. Her temperature was 37.1 °C (98.7 °F), her pulse 110 per minute, and her blood pressure 130/70 mm Hg. Her pupils were fixed and dilated, and she had no corneal, gag, or deep-tendon reflexes. The serum sodium level was 140 mmol per liter, the potassium level 6.6 mmol per liter, the chloride level 110 mmol per liter, and the bicarbonate level 1 mmol per liter (anion gap, 29 mmol per liter). The serum creatinine concentration was 2.8 mg per deciliter (248 μmol per liter), and the lactate concentration 10.1 mmol per liter. The osmolar gap (the difference between the measured value and the predicted value) was 84 mOsm per kilogram. Urinalysis revealed calcium oxalate crystals. The serum concentration of ethylene glycol was 2600 mg per liter; no other toxins were detected. Serial arterial-blood gas values are shown
Two hours after admission, hemodialysis was begun and continued intermittently for 48 hours. To reduce the conversion of ethylene glycol to its acid metabolites, ethanol was added to the dialysate1 and then given by gavage in a dose of 600 mg per kilogram of body weight, followed by intragastric infusion at a rate of 200 mg per kilogram per hour for 37 hours. Renal failure did not develop, and the patient recovered completely.
Although the arterial-blood pH of normal subjects may fall to 6.80 after extreme exertion,2 in a clinical setting such a level is usually fatal. Arterial-blood pH values of 6.78, 6.57, and 6.49 have been reported in patients who survived poisoning with ammonium chloride3 and strychnine4 and isoniazid overdose,5 respectively.
85-YO male in ED
good article on medical response to Boston Marathon bombing
Drs. Voyles, Busby, Spaulding
85-YO male in ED
Diagnosis of COPD should be supported by objective data (e.g., PFT)
unable to assess CVP by JVD in obese patients
Cold upper and lower extremities; slight mottling of lower extremities = vasoconstriction
2/6 early SEM at the base only - could be due to turbulence from tachycardia, anemia, sclerotic valve leaflets
gallop uncertain - hard to tell at high heart rates S3 and S4 may blend with shortened diastolic period. also hard to hear due to low pitch. S4 occurs with atrial kick and LV hypertrophy from chronic HTN
Shallow respirations and decreased breath sounds in both bases; coarse rhonchi in all lung fields; scattered expiratory wheezes; no rales - rhonci = gunk in airways; shallow sounds = low volume breaths; expiratory wheezing could be airway sounds or "cardiac asthma" where edema in lymphatics constricts airways = wheezing. no rales or crackles could be due to shallow breaths and no opening of collapsed airways. may become audible if pt. sits up and takes a big breath.
not making urine - maybe he voided in ambulance or is not making urine. BPH may contribute. ??? not making urine vs. not voiding. hypotension responsible?
Place Foley catheter and maybe bedside bladder scan.
Echocardiograms hard to obtain with morbidly obese pts. transesophageal echo is a way around this.
don't give nl saline and furosemide at the same time. need more data; e.g., blood pressure (via arterial line with pressure transducer in obese pts. since cuffs aren't accurate) saline may increase
BP BUT worsen pulmonary edema.
Flank pain presenting complaint - don't drift far from this; e.g., AAA
no abdominal tenderness since abdominal pain is peritoneal and AA is in peritoneal space.
CT scan problematic with high Cr. Do it anyway if suspicion is AAA.
PE vs. AAA - in one case heparinize not in AAA (would kill him)
Calcification occurs with smoking, obesity, and HTN
PE does not present with back and flank discomfort but AAA that ruptures does.
Tests for AAA
Drs. Voyles, Busby, Spaulding
85-YO male in ED
Diagnosis of COPD should be supported by objective data (e.g., PFT)
unable to assess CVP by JVD in obese patients
Cold upper and lower extremities; slight mottling of lower extremities = vasoconstriction
2/6 early SEM at the base only - could be due to turbulence from tachycardia, anemia, sclerotic valve leaflets
gallop uncertain - hard to tell at high heart rates S3 and S4 may blend with shortened diastolic period. also hard to hear due to low pitch. S4 occurs with atrial kick and LV hypertrophy from chronic HTN
Shallow respirations and decreased breath sounds in both bases; coarse rhonchi in all lung fields; scattered expiratory wheezes; no rales - rhonci = gunk in airways; shallow sounds = low volume breaths; expiratory wheezing could be airway sounds or "cardiac asthma" where edema in lymphatics constricts airways = wheezing. no rales or crackles could be due to shallow breaths and no opening of collapsed airways. may become audible if pt. sits up and takes a big breath.
not making urine - maybe he voided in ambulance or is not making urine. BPH may contribute. ??? not making urine vs. not voiding. hypotension responsible?
Place Foley catheter and maybe bedside bladder scan.
Echocardiograms hard to obtain with morbidly obese pts. transesophageal echo is a way around this.
don't give nl saline and furosemide at the same time. need more data; e.g., blood pressure (via arterial line with pressure transducer in obese pts. since cuffs aren't accurate) saline may increase
BP BUT worsen pulmonary edema.
Flank pain presenting complaint - don't drift far from this; e.g., AAA
no abdominal tenderness since abdominal pain is peritoneal and AA is in peritoneal space.
CT scan problematic with high Cr. Do it anyway if suspicion is AAA.
PE vs. AAA - in one case heparinize not in AAA (would kill him)
Calcification occurs with smoking, obesity, and HTN
PE does not present with back and flank discomfort but AAA that ruptures does.
Tests for AAA
- KUB xray
- aortagram
- CT scan -
AAA can create ATN if renal arteries are involved.
Huge surgical risks with this pt. (not a candidate for a haircut)
option = endovascular repair
pt. died on operating table with refractory shock.
Tuesday, April 30, 2013
Pediatric Nephrology
Pediatric Nephrology
1. Renal Tubular Acidosis
• Hyperchloremic, Non-Anion Gap, Metabolic acidosis
– Alkali loss: Intestine or Kidney?
• What should the kidney do in Acidosis?
– Secrete acid and retain alkali.
• Acid urine (U pH < 5.5)
• If the kidney is not excreting acid and retaining HCO3 then something is amiss?
Proximal RTA (type 2)
– Proximal Tubule
– HCO3 re-absorption is Dysfunctional
.
Proximal RTA
Impaired proximal tubule (PT) HCO3 absorption
• HCO3
reabsorption in PT is decreased
• Resulting in “Extra HCO3” in the urine (FeHCO3>5%)
• Serum HCO3 decreased
•Urine pH
• Low in acidosis
• High with treatment
• Distal RTA (type 1 & 4)
– Distal Tubule
– H+ secretion is Dysfunctional
Distal RTA type 1
Impaired Distal Tubule acid secretion
• Hydrogen ion secretion in the Distal Tubule is impaired.
• K+ reabsorption is impaired.
• H+ is not available to convert NH3 to NH4
+
• Limited acid excretion and new HCO3 creation.
• Urine pH is always high (>5.5)
Urine pH in RTA - not useful in diagnosing RTA
Can be High or Low
• Proximal RTA
– When Plasma HCO3 is low, the impaired proximal tubule can handle the reabsorption load resulting in an:
– Acidic urine.
– When plasma HCO3 is normalized (with treatment), the impaired proximal tubule cannot handle the filtered load resulting in an:
– Alkaline Urine.
• Distal RTA
– Final acidification of the urine occurs in the distal tubule via H+ secretion. This is defective in Distal RTA and hence:
– Urine pH is always aAkaline
Urine Anion Gap:
Urine (Na+ + K+) - Cl-
• Useful to differentiate the type of RTA
– It tells us about NH4
+ excretion:
U AG = Na+ + K+ + NH4 + = Cl- + HCO3
-
• Urine AG is Negative in Proximal RTA or healthy kidney
• Urine AG is Positive in Distal RTA.
• U AG is not accurate if:
– Dehydration, unmeasured ions are present (ketoacidosis) or
diuretics are in use.
How to Diagnose RTA?
• RTA likely exists when you have:
– Hyperchloremic, non-anion gap, metabolic acidosis.
– No extra-renal (i.e. GI) HCO3 losses
– No excessive intake of a Cl- containing acid
• Urine pH and Urine anion gap are useful to
differentiate the type of RTA.
Why do we care about RTA?
• Chronic acidosis leads to:
– Polyuria, failure to thrive and growth delay.
• Chronic hypokalemia causes:
– Nephrogenic D.I., urinary retention, ileus, muscle weakness / rhabdomyolysis and rarely cardiac arryhmias.
• Calciuria in Distal RTA leads to:
– Rickets and Kidney Stones.
• Associations with structural, auto-immune or metabolic disease:
– Obstructive Uropathy
– Cystinosis, Glycogen storage disease, Lowe’s syndrome, DM I & II
– Sjogren syndrome, Hypoaldosteronism
– Etc.
Management of RTA
1. Oral alkali: 1- 5 meq/kg/day*
– Na-Bicarbonate
– K- Citrate
*Alkali needs lessen with age
2. Evaluate and treat associated conditions.
Classification of RTA
• Proximal RTA (type 2)
– Dysfunction of proximal tubule re-absorption of filtered HCO3 .
– Often seen with generalized proximal tubular dysfunction
• Fanconi Syndrome: wasting of K+, HCO3, AAs, glucose, LMW protein, etc.
– Clinical Association: Cystinosis and Lowe’s Syndrome.
• Distal RTA (type 1 & 4)
– Dysfunction of distal H+ secretion and Ammonium (NH4+) generation
– 2 types:
• Type 1 - Low K+
• Type 4 - High K+ (due to lack of aldosterone activity)
– Clinical Association: Obstructive Uropathy, Hypoaldosteronism, Drugs or Chronic renal failure.
Metabolic Alkalosis and Hypokalemia
- Tubular Disorders
- Bartter’s Syndrome
- Defect in the Furosemide -sensitive distal Na-K-2Cl Transporter:
- • Salt (NaCl) wasting => polyuria, dehydration=> Renin-angiotensinaldosterone activation => Hypokalemic Alkalosis.
- • Birth Hx: Polyhydramnios
- • S/S:
- – Dehydration, constipation, vomiting, muscle weakness.
- – Polydipsia, polyuria, salt craving
- – Urine Calcium wasting
- – Failure to thrive and short stature
- – Hyper-Reninemia, but NO HTN
- Gitelman Syndrome
- Similar to Bartter’s, but milder.
- Hypokalemic Metabolic Alkalosis.
- Low –Magneseum level.
- Defect in the Thiazide sensitive Na-Cl transporter.
- High Renin, but no hypertension. (increase in vasodilatory PGs)
- May present later in life with fewer symptoms
- Liddle Syndrome
- hypokalemic alkalosis plus HTN
- Primary increase in collecting duct Na+ reabsorption and K+ secretion.
- Hyper-Aldosteronism -
- hypokalemic alkalosis plus HTN
- Persistent Vomiting
- hypokalemic alkalosis
- Self-Induced Metabolic Alkalosis
- • Surreptitious Vomiting or Diuretics
- – Anorexia / Bulemia
- • Dental erosions, calluses or ulcers on dorsum of hand, puffy cheeks (salivary gland hypertrophy).
- – Urine Cl-
- • Low with chronic vomiting or prior diuretics
- • High with diuretics or Bartter / Gitelman
- – Check Urine diuretic levels if needed.
- Diuretic abuse
Monday, April 29, 2013
Chronic Kidney Disease
Chronic Kidney Disease
Modifiable risk factor
Creatine levels difficult to interpret
example:
At What Level of Creatinine Does a 65-Year-Old Diabetic, Hypertensive White Woman Weighing 50 Kilograms Have CKD?
• 77% said:
Creatinine > 1.5 mg / dl
– GFR = 37 mL/min/ 1.73 m2
– Ccreat = 30 mL/min
BUT
• Creatinine = 1.0 for GFR = 59 mL/min/1.73 m2
The Risk of Kidney Failure is Not Uniform
Relative risks compared to Whites:
African Americans 3.8 X
Native Americans 2.0 X
Asians/Pacific Islander 1.3 X
The relative risk of Hispanics compared to non-Hispanics is about 1.5 X
Risk factors for CKD
Nonmodifiable risk factor
- Older age
- Black Race
- Genotype
- Prematurity
Modifiable risk factor
- Hypertension
- Proteinuria
- Dyslipidemia
- Hyperuricemia
- Smoking
Mechanisms of Kidney Disease Progression
• Adaptive changes lead to maladaptive consequences
• Hypertension
• Hyperfiltration
– Elevated glomerular pressure
• Glomerular growth
– Increased wall stress
• Increased ammoniagenesis
• Complement activation and tubulo-interstitial disease
There are ~12 million adults in the US with CKD 3 or greater, but only ~500,000-600,000 patients with ESRD or a kidney transplant.
Why the disparity????
• Far more CKD patients die due to CVD than reach ESRD
• CKD is considered by some to be a coronary equivalent
– Data indicate that patients with lower GFRs (<45ml/min) and microalbuminuria or proteinuria carry a very high CVD risk (~ equivalent to a prior history of coronary disease).
Risk Factors for CVD in CKD
• Age
• Diabetes mellitus
• Smoking
• Hypertension
• Dyslipidemia
• Physical inactivity
• Menopause
• Obesity
• Anemia
• Hyperparathyroidism
• Hyperphosphatemia*
• Hypocalcemia
• Effects of dialysis
• Hypoalbuminemia and malnutrition
• Systemic inflammation
• Hyperhomocysteinemia
• Volume overload
UREMIA = urine in the blood
Uremia as a Clinical Syndrome
• Renal excretory failure
• Retained products of metabolism
• Related to protein intake
• Partially dialyzable
• Exact nature is unknown
• E.g. Small molecules (Urea etc.), lipid soluble molecules, middle molecules – urea (BUN), hormones, polyamines, middle molecules, serum proteases, trace elements, pyridine derivatives, 2-microglobulin
• Loss of metabolic and endocrine functions normally performed by the intact kidney
Uremia: Common Symptoms
• GI Nausea, vomiting, diarrhea
• CVS Dyspnea, edema, chest pain
• Neuro Restless legs, twitching, confusion
• Skin Pruritus, bruising, uremic frost
• MSK Bone pain, arthritis
Uremia: The Common Signs
• Sallow pallor, bruising
• Uremic fetor
• Hypertension
• Pericardial rub
• Alteration of consciousness
• Neuropathy
Alonzo Morning _NBA star FSGS
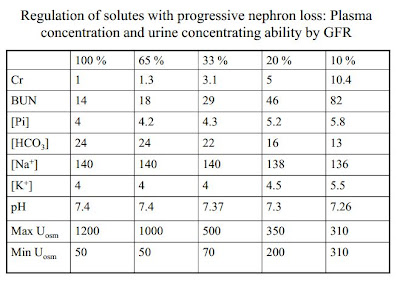
Adaptation of kidney function
Mechanisms of Adaptive Natriuresis in CKD
• Signal: ECF volume expansion
• Potential effectors
– Atrial natriuretic peptide (ANP)
– Other circulating natriuretic factors
– Local renal vasoactive factors
Clinical Manifestation of Sodium Balance in CKD
• Common ("Hoagie Night")
– Weight gain
– Peripheral edema
– Pulmonary edema
• Uncommon
– Renal Na wasting (ECF volume depletion)
– Weight loss
– Systemic hypotension
Potassium Homeostasis
Renal K Handling in CKD
- • K balance is very well maintained with progressive nephron loss
- • Filtered load of K decreases as GFR falls
- • K reabsorption is similar in normal and diseased kidneys
- • Adaptation: Distal K secretion is increased in proportion to the decrement in GFR
- via aldosterone
Mechanisms of Adaptive K Secretion in CKD
• Enhanced Na,K-ATPase activity
– Extracellular K concentration
– Aldosterone
• Increased distal tubular flow
– Adaptive natriuresis
– Osmotic diuresis per nephron
– Chronic metabolic acidosis
Hyperkalemia in CKD
• Aldosterone-related
– Hypoaldosteronism
• Idiopathic (Diabetes Mellitus)
• ACE inhibitors
– Pseudohypoaldosteronism
• K-sparing diuretics
• Distal flow-related
– ECF volume depletion
– Congestive heart failure (without diuretics)
• Insulin-related
– Diabetes mellitus
– Fasting
– Malnutrition
• Miscellaneous
– B-adrenergic agonists (esp. in diabetics)
– Dietary indiscretion
Anemia and Kidney Failure
Primary factors
- Relative erythropoietin deficiency
- Shortened rbc lifespan
- “uremic toxins”
- EPO may be a red blood cell “survival factor”
- Inhibitors of erythropoiesis--”uremic toxins”
Features of the anemia of CKD:
- Normocytic and normochromic
- Low reticulocyte count
- Normal bone marrow--and usually not needed to diagnose
- Serum erythropoieitin level low-normal--not needed to diagnose
Treatment of CKD Anemia
- Give EPO?
- but normalizing Hb leads to higher rate of death in CKD patients
- cut off is Hb or 7-8 g/dL
*Phosphate Homeostasis
coronary calcifications occur with high PO4 x Ca
Phosphate Homeostasis review articel
Bone/Ca/Phos/PTH Management
- • Low Phosphate diet
- • Maintain Ca X Phos product less than 55mg2/dl2
- • Phosphate binders with meals
- positive charge binders; e.g., calcium (Tums with meals)
- Use of Calcitriol or D3 analogues
- dampens stimulation of PTH
- • Monitor Ca, Phos, PTH
- • Avoid the use of aluminum
- neurotoxic
Subscribe to:
Posts (Atom)